Note where the line crosses the methane axis.Connect the points with a straight line.On the right-hand vertical axis, locate and mark the point containing the temperature 60☏.On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia.

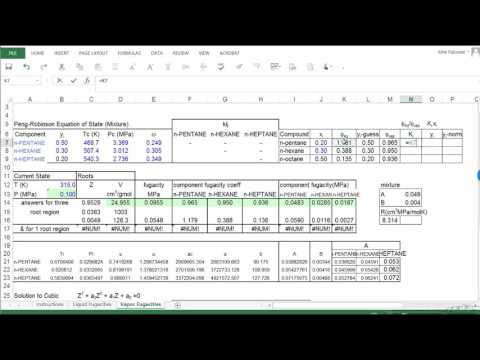
Example įor example, to find the K value of methane at 100 psia and 60 ☏. Many DePriester charts have been printed for simple hydrocarbons. temperature, and K-factor for various light and heavy hydrocarbons. from Introduction to Chemical Engineering Thermodynamics (7th ed) by Smith, J.M., Van. DePriester charts Source: A Dictionary of Chemical Engineering. K (or DePriester) Chart (high T range) in American Engineering Units from Introduction to Chemical Engineering Thermodynamics (7th ed) by Smith, J.M., Van Ness, H.C., and Abbott, M.M. McWilliams (McWilliams, 1973) fitted these charts to the following polynomial equation (5) ln K a T 1 T 2 + a T 2 T + a T 3 + a p 1 ln p + a p 2 p 2 + a p 3 p where T in R and p in. pressure and temperature that are valid up to around 6000 psi pressures or more. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. K (or DePriester) Chart (low T range) in American Engineering Units. DePriester (DePriester, 1953) presented K-value charts for light hydrocarbons vs.

These nomograms have two vertical coordinates, one for pressure, and another for temperature. DePriester in an article in Chemical Engineering Progress in 1953. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.
